Light the Way
Ahmed Abdelfattah's Voltron uses a light-sensing protein to convert the brain's subtle voltage changes into a readout that makes the activity of neurons visible.
By Gretchen Schrafft, Science Communications Specialist
WHEN WE THINK of voltage, we don’t tend to think of brain cells. Yet a single neuron fires an average of a million electrical signals a day to help drive brain function. These signals are infinitesimal –– just 0.1 volts. (Compare this to 12 volts of a car battery or 100 million volts in a lightning strike.) To understand brain function, neuroscientists need to be able to measure this essential electrical activity that allows neurons to communicate.

But traditional methods have been unable to measure voltage changes without damaging the cells, or to pinpoint the activity to specific neuron networks.
Enter Ahmed Abdelfattah, Robert J. and Nancy D. Carney University Assistant Professor of Brain Science and an assistant professor of neuroscience. Abdelfattah was nearing completion a chemistry Ph.D., looking at protein structure and function, when he first learned about this barrier faced by neuroscientists. He thought: “What if I could use a light-sensitive protein to convert cell membrane voltage into a readout we could actually see?”
For the next five years, then a postdoc at HHMI Janelia Research Campus, Abdelfattah worked with a team of collaborators to methodically coax a protein found in a type of light-sensing algae to evolve: enhancing its optical properties so that it would change predictably in relation to voltage, and further brightening those properties by linking the protein to an ultra-bright synthetic fluorescent dye. By 2019 Abdelfattah had created a voltage sensor that he christened Voltron, named after a 1980s cartoon robot whose multiple colors are like the multiple dyes the tool can use.
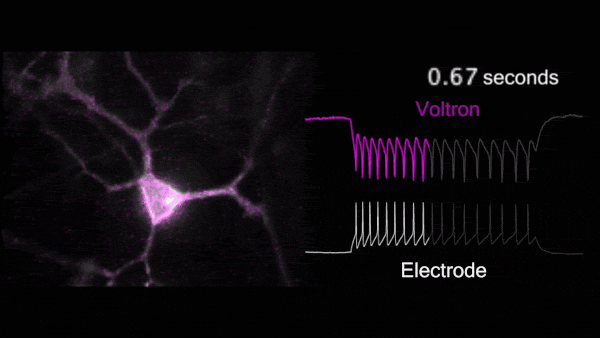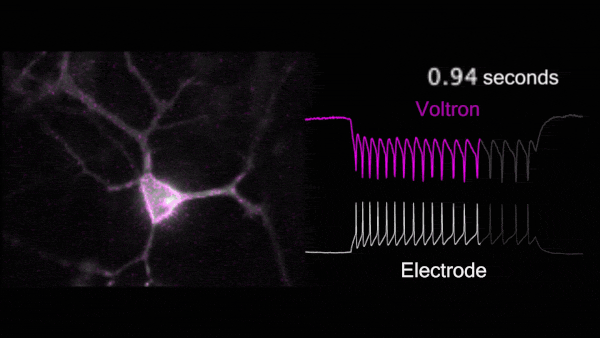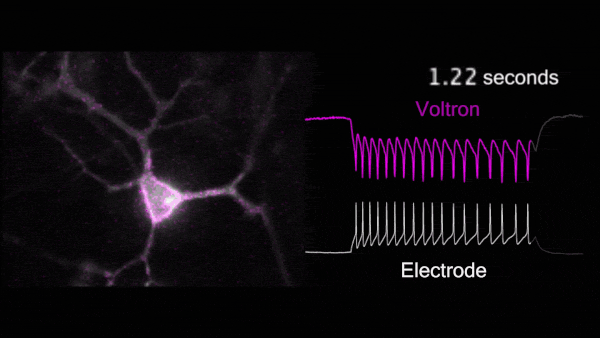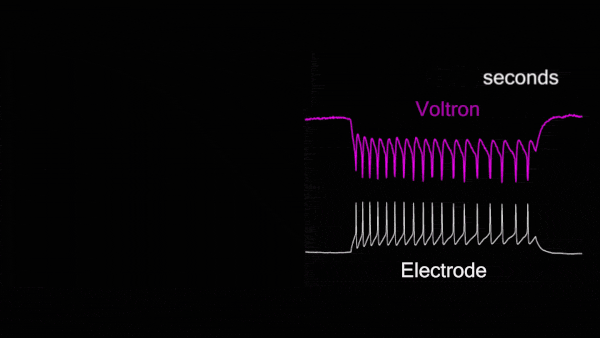
For the first time, Voltron made possible non-invasive, hourlong, real-time video recordings of neurons firing in awake, behaving animals. Because Voltron is genetically encoded, scientists can target and record from specific neurons they want to observe, as opposed to capturing activity from large regions of the brain.
Compared to traditional techniques, using Voltron is “like watching the movie of what the brain's actually doing, instead of trying to listen to it through a tin can,” said Chris Moore, Adam and Margaret Korn Professor of Brain Science and a collaborator of Abdelfattah. “Ahmed has made a way to watch the movie of the brain's dynamics in real time.”
_______________
For the first time, Voltron made possible non-invasive, hourlong, real-time video recordings of neurons firing in awake, behaving animals
_______________
He has since followed the original Voltron with Voltron 2, an upgraded version that’s much more sensitive to voltage changes. He continues to advance the technology; his laboratory is currently teeming with dozens of developing variants, each designed to illuminate different aspects of brain behavior.
“One reason there are so many unanswered questions about the brain is because the right tools don’t exist,” said Abdelfattah. “My main driving force is to put our tools in the hands of people working to answer new questions that couldn’t be answered before.”
The power of collaboration
ALEXANDER FLEISCHMANN, Provost’s Professor of Brain Science and a professor of neuroscience, studies the sense of smell. Smell is tightly linked to learning and memory, and odor tests are increasingly administered as a first line of assessment for dementia. To learn how neurons form odor memories, Fleischmann needed a better way to view the olfactory cortex deep in the brain –– but it was too far for the green and yellow light of Voltron to penetrate.
Red light, however, can travel farther than green. As Abdelfattah arrived at Brown, he began developing variants of Voltron to use red dyes. Fleischmann’s team tested the variants in their experiments and provided critical user feedback that helped Abdelfattah create a red-light Voltron that could give the Fleischmann lab a much richer picture of what happens in the brain the instant an animal smells an odor.
Just as their collaboration helped inform a new iteration of Voltron, it has also inspired new research questions. “Our conversations now drive our experimental design,” Fleischmann said. “It’s moved from, ‘We have an experiment –– will your tool work?’ to, ‘With the tools you have, how can we design an experiment for the questions we have?’”
IN THE SUMMER of 2023, Michael Hoppa was commiserating with colleagues about a problem he was running into in his research.
Hoppa co-directs the Neurobiology course at the Marine Biological Laboratory in Woods Hole, Massachusetts, a summer neuroscience program for researchers to learn advanced techniques in physiology and microscopy. Abdelfattah, an alum of the program, was the first faculty member Hoppa recruited after becoming co-director.
He wanted to study human genetic mutations that he suspected changed the way pores in the membranes of neurons open and close to regulate the flow of electrical signals, contributing to conditions like epilepsy. To measure voltage at a super-low level of .001 volts –– a single millivolt –– the gold-standard is a technique called patch clamping. But this involves sealing a glass micropipette to a neuron’s membrane, an invasive approach that ultimately alters or kills the neuron. Patch clamping also isn’t practical for measuring the ion channels (which help conduct electrical signals) in neuronal axons deep in the brain, or for measuring more than one neuron at a time.
Inspired, Abdelfattah started tinkering with Voltron and realized he could fine-tune the molecule's sensitivity to pick up the subtle voltage shifts that occur even when a neuron is at rest. Suddenly, and for the first time ever, here was a sensor detecting changes at this single-millivolt level. Such a sensor could answer questions not just about epilepsy but also about tiny, yet critical, nuances of many different disease states.
When Hoppa and Abdelfattah met back up again in summer 2024, they paired the new sensor together with an ultra-fast microscope. Through a prestigious award in 2025 from the Keck Foundation, they are developing their technology for a calibrated voltage sensor, with Hoppa building the specialty microscopes needed for the project.
Shaping the Future
BY MAKING VOLTAGE changes visible, Abdelfattah and his collaborators are mapping the path toward new understandings of brain dynamics and treatments for disease –– visualizing brain communication at cellular resolution and millisecond timescales. The reach of Voltron is ever-growing: Currently, 50 to 100 labs collaborate with Abdelfattah, and more than 1,000 use Voltron and its variants.
But Hoppa emphasizes that Abdelfattah’s impact is notable in another impressive metric: the Neurobiology summer students who gravitate toward him as a teacher.
“He’s captured the attention of the next generation of scientists,” said Hoppa. “They see his approach as the future.”
